New Radio-Theranostic Promises to Broaden Targeted Radiation in Solid Tumors

ONCOLife |
4 April 2026
Researchers at UT MD Anderson developed MIL33B, a high-affinity antibody that selectively targets the tumor-associated 4Ig-B7-H3 isoform while sparing the soluble decoy form. In preclinical models, radioligand therapy with 90Y-MIL33B achieved 53% long-term survival and triggered CD8+ T-cell activation with immune memory. The humanized version, BetaBart, has now entered a Phase I/II clinical trial.
Targeted radiotherapy has transformed care for a limited number of cancers, but its reach remains narrow. A new preclinical study from researchers at The University of Texas MD Anderson Cancer Center suggests that may begin to change. Their work describes a novel antibody, MIL33B, designed to distinguish between two closely related forms of B7-H3, a protein widely overexpressed in solid tumors and long viewed as an appealing, but frustratingly difficult, therapeutic target.
In humans, B7-H3 exists in two isoforms. The 4Ig-B7-H3 form is enriched on tumor cells, while the 2Ig form circulates in the bloodstream as a soluble decoy. Therapies that fail to discriminate between the two risk reduced tumor targeting and greater off-target effects.
“We’ve been working on this for almost a decade,” said Dr. David Piwnica-Worms, chair at Cancer Systems Imaging, UT MD Anderson Cancer Center. “The ability to effectively target the B7-H3 protein has been an attractive but elusive goal for scientists since the early days of immune checkpoint therapies. This is a very exciting publication because we present an antibody that can precisely target the types of B7-H3 proteins that are found on cancer cells, but avoid the ones found in the blood stream.”
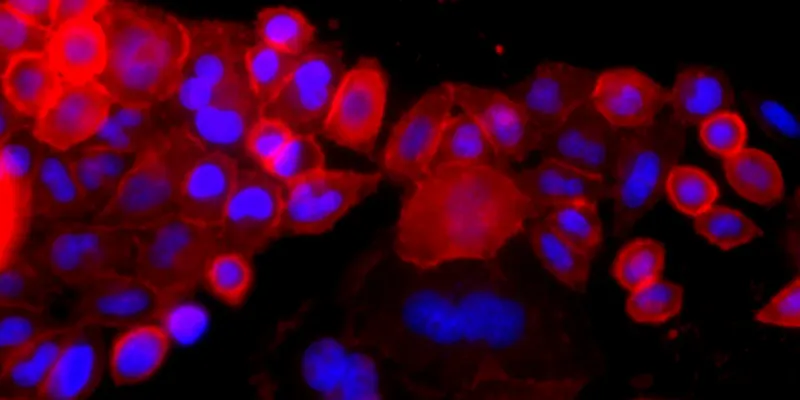
According to the Theranostics paper, MIL33B binds the tumor-associated 4Ig-B7-H3 isoform with very high affinity, about 72 picomolar, and shows 8- to 18-fold selectivity over the soluble 2Ig form. In laboratory testing, the antibody localized strongly to the cell membrane. In animal models, PET-CT imaging with zirconium-89 labeled MIL33B confirmed robust and selective tumor uptake across multiple cancer settings, including cervical, breast, melanoma, and colorectal models engineered to express human 4Ig-B7-H3.
That imaging capability is only one part of the story. The more consequential application is therapeutic. By linking the antibody to a beta-emitting radioisotope, the team created a radioligand approach that delivers tumor-directed radiation while sparing normal tissue more effectively than conventional systemic therapy. In the published study, a single systemic dose of yttrium-90 labeled MIL33B produced long-term survival in 53% of mice in an otherwise fatal colorectal tumor model, and that effect depended on 4Ig-B7-H3 expression.
The treatment did not appear to act solely as a radiation payload. Immunologic analyses showed activation of CD8-positive T cells and evidence of immune memory, suggesting the therapy may also prime an antitumor immune response.
“When we challenged previously treated tumor models that had responded to initial treatment, we saw that most did not develop new tumors,” Dr. Piwnica-Worms said. “Initiating a lasting immune response, termed immune memory, from a radio-theranostic would be a significant achievement because it would mean these drugs could have a long-term benefit for patients even after initially attacking the tumor itself.”
That possibility is especially important in a field where approved radio-theranostics remain confined to relatively specific indications, such as PSMA-targeted treatment in selected prostate cancers. A B7-H3-directed platform could potentially extend this strategy to a much broader range of solid tumors, particularly aggressive cancers with limited targeted options.
The MD Anderson group’s preclinical antibody has now been translated into a humanized clinical candidate known as BetaBart. Developed through Radiopharm Ventures, a joint effort between UT MD Anderson and Radiopharm Theranostics, the agent is already being evaluated in a Phase I/II trial.
If the clinical data support the preclinical promise, BetaBart could mark an important step toward expanding radiotheranostics beyond their current boundaries, not only by hitting tumors more precisely, but by helping the immune system remember them.










Comments
No Comments Yet!