Reengineering CAR-T Cells to Last Longer in the Fight Against Cancer and HIV

ONCOLife |
15 March 2026
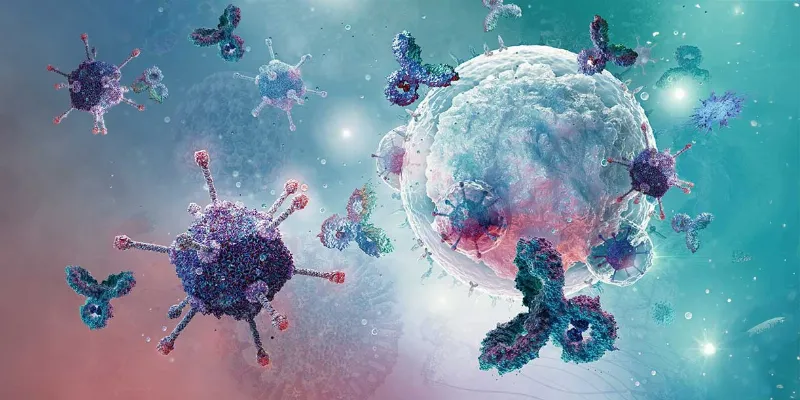
Researchers developed a new CAR-T manufacturing strategy using the cytokine fusion scaffold HCW9206 to generate longer-lasting immune cells. This approach produced CAR-T cells enriched in T memory stem cells (>50% vs <5%), demonstrating enhanced persistence, stronger recall responses, sustained control of blood cancers, and improved HIV suppression in preclinical models.
Chimeric antigen receptor T-cell therapy has transformed cancer treatment by turning a patient’s own immune cells into targeted tumor killers. Yet despite dramatic initial responses, the long-term durability of these engineered cells remains a major limitation. Over time, many CAR-T cells lose their functional potency, allowing disease to return.
A research team led by scientists at Albert Einstein College of Medicine has developed a new strategy to overcome this obstacle. In a study published in Science Advances, the team describes a new manufacturing method that produces CAR-T cells with substantially greater persistence and regenerative capacity. In mouse models, the engineered cells maintained anti-tumor and antiviral activity far longer than those produced through conventional techniques.
A durability problem in cellular therapy
CAR-T therapy works by extracting T cells from a patient, genetically modifying them to express receptors that recognize disease targets, and reinfusing them into the bloodstream. These reprogrammed cells then seek out and destroy cancerous or virus-infected cells.
Although the approach has produced striking remissions in blood cancers, its effectiveness often fades. In roughly half of treated patients, declining CAR-T activity allows tumors to re-emerge. The same biological limitation has hindered efforts to apply CAR-T technology to chronic viral infections such as HIV.
"Our goal was to engineer therapeutic immune cells so they would not only be powerful killers but also long-lived and capable of self-renewal, to markedly extend their effectiveness after infusion into patients," said Dr. Harris Goldstein, senior author of the study and professor of pediatrics and immunology at Einstein. “By improving how we generate CAR-T cells, a treatment that acts as a 'living drug,' we would prolong their functional activity and prevent disease relapse after their potency wanes.”
Building longer-lived immune cells
Conventional CAR-T manufacturing relies on anti-CD3/CD28 stimulation to activate T cells during production. While effective at generating large numbers of cells, this process often drives them toward a terminally differentiated state, reducing their lifespan once infused. To address this, the Einstein team developed an alternative activation strategy using a multi-cytokine fusion scaffold known as HCW9206. The engineered protein links three cytokines, IL-7, IL-15, and IL-21, signaling molecules known to promote T-cell survival and immune memory.
Instead of the traditional activation protocol, the fusion scaffold stimulates T cells during CAR-T production. The result is a markedly different cell population. More than half of the resulting CAR-T cells display characteristics of T memory stem cells, a rare subset capable of self-renewal and long-term persistence. By contrast, fewer than 5 percent of CAR-T cells generated using the standard method exhibit this stem-like phenotype.
“T memory stem cells are considered to be critical for long-term immune persistence,” said Dr. Goldstein. “They can continually replenish the pool of active CAR-T cells, a crucially important attribute for their long-term success in combating both cancer and HIV infection.”
Stronger recall responses against disease
The biological impact of this manufacturing shift became clear in disease models. In a mouse model of human leukemia, both standard and scaffold-generated CAR-T cells initially eliminated cancer cells. However, when researchers later reintroduced leukemia cells to simulate relapse, only the scaffold-derived CAR-T cells expanded again and prevented tumor recurrence.
The same durability advantage appeared in a humanized mouse model of HIV infection. CAR-T cells produced using the HCW9206 scaffold eliminated significantly more HIV-infected cells and suppressed viral replication more effectively than those produced through conventional methods.
Implications for next-generation cellular therapy
The findings suggest that CAR-T performance may depend as much on how the cells are manufactured as on the genetic engineering itself. By enriching for stem-like T cells capable of long-term renewal, the cytokine scaffold platform may allow engineered immune cells to remain active for years.
“Now that we have shown that we can generate potent CAR-T cells that are longer-lived, we may be able to reduce blood cancer relapse rates and improve long-term remission for cancer patients,” Dr. Goldstein said. “For HIV, immune cells with this kind of staying power may one day help maintain viral control after stopping antiretroviral therapy, a critical step toward sustained drug-free remission and, potentially, a functional cure.”
If confirmed in clinical studies, the approach could mark an important step toward more durable cellular immunotherapies, expanding the potential of CAR-T technology beyond short-term tumor control to long-term disease management.





Comments
No Comments Yet!