Vitamin B2’s Hidden Role in Cancer Survival May Open a New Therapeutic Target
14 March 2026
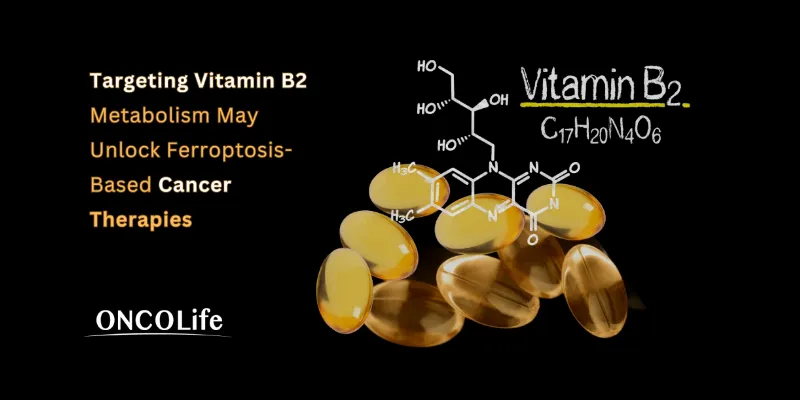
A Nature Cell Biology study shows that vitamin B2 metabolism helps cancer cells evade ferroptosis by stabilizing ferroptosis suppressor protein-1 and sustaining antioxidant defenses. Disrupting this pathway, including with the riboflavin antimetabolite roseoflavin, sensitized tumor cells to ferroptosis, highlighting vitamin B2 metabolism as a promising therapeutic target in cancer.
Vitamin B2, long recognized for supporting cellular metabolism, may also help cancer cells evade death. Researchers at the Rudolf Virchow Centre at Julius-Maximilians-Universität Würzburg report that riboflavin, better known as vitamin B2, plays a central role in protecting tumor cells from ferroptosis, an iron-dependent form of programmed cell death. Their findings, published in Nature Cell Biology, suggest that disrupting riboflavin metabolism could become a new strategy to weaken tumors and improve cancer treatment.
Ferroptosis has attracted increasing interest in oncology because it represents a distinct mechanism of cell death driven by iron-mediated lipid peroxidation. Unlike apoptosis, ferroptosis occurs when oxidative damage to membrane lipids overwhelms a cell’s antioxidant defenses. Many tumors evade this process by reinforcing redox protection systems that neutralize oxidative stress.
“Vitamin B2 plays a crucial role in protecting cancer cells from ferroptosis, a special form of programmed cell death,” says PhD student Vera Skafar from the research group led by José Pedro Friedmann Angeli, Professor of Translational Cell Biology.
The new study identifies riboflavin metabolism as a key component of this protective network. Through a focused CRISPR–Cas9 screen, the researchers found that riboflavin-derived cofactors help stabilize ferroptosis suppressor protein 1 (FSP1), a critical inhibitor of ferroptosis. FSP1 protects cellular membranes by regenerating lipophilic antioxidants such as ubiquinone and vitamin E, thereby preventing damaging lipid peroxidation.
The link between riboflavin and FSP1 lies in flavin cofactors produced through the riboflavin–FMN–FAD metabolic pathway. In particular, the cofactor flavin adenine dinucleotide (FAD) stabilizes the FSP1 protein and maintains its enzymatic activity. When riboflavin metabolism is disrupted, FSP1 becomes unstable and is targeted for degradation through the ubiquitin–proteasome system, ultimately reducing the cell’s ability to resist oxidative damage.
This dependency creates a vulnerability. In experiments using genome editing and cancer cell models, riboflavin deficiency markedly increased ferroptosis sensitivity. The results suggest that targeting riboflavin metabolism could remove an important protective shield that tumors rely on for survival.
To explore this possibility, the Würzburg team investigated roseoflavin, a bacterial compound structurally similar to riboflavin. Acting as an antimetabolite, roseoflavin interferes with the riboflavin metabolic pathway and disrupts FSP1 activity. In laboratory experiments, the compound induced ferroptosis in cancer cell models at low concentrations.
Professor Friedmann Angeli explains the significance of this observation: “It turned out that roseoflavin triggers ferroptosis in low concentrations,” he says, adding that “our experiments show the feasibility of this concept.”
The findings highlight riboflavin metabolism as a previously underappreciated regulator of ferroptosis resistance. The work also reveals a broader biological principle. Just as selenium availability controls the activity of glutathione peroxidase 4 (GPX4), another ferroptosis regulator, riboflavin availability determines the stability and function of FSP1. In both cases, micronutrients influence the activity of critical antioxidant enzymes, shaping how cells respond to oxidative stress.
The study also raises questions about how laboratory conditions may influence ferroptosis research. Standard cell culture media often contain riboflavin concentrations far higher than those found in human plasma.
When the researchers tested cells under more physiological riboflavin levels, they observed significant differences in FSP1 expression and ferroptosis susceptibility. This suggests that nutrient availability could strongly influence experimental outcomes and possibly clinical responses.
Although roseoflavin itself is not yet a therapeutic drug, the concept of targeting riboflavin metabolism is gaining attention. Unlike direct inhibitors of FSP1, riboflavin antimetabolites act upstream in a metabolic pathway that cells depend on for essential cofactor production. This could make it more difficult for cancer cells to develop resistance.
The Würzburg group is focusing on identifying and optimizing inhibitors of riboflavin metabolism and evaluating them in preclinical cancer models. If successful, such agents could become part of a new class of therapies designed to trigger ferroptosis selectively in tumor cells.
Ferroptosis itself is not limited to cancer biology. As Professor Angeli notes, “Ferroptosis is not only relevant to cancer. Increasing evidence suggests that it also contributes to pathological processes in neurodegenerative diseases and in tissue damage following organ transplantation or ischemia-reperfusion injury.”
Understanding how vitamin metabolism shapes ferroptosis may therefore have implications far beyond oncology. By revealing how a common micronutrient controls one of the cell’s most important survival pathways, the study opens a new line of investigation into how metabolism, nutrition, and redox biology intersect in disease.







Comments
No Comments Yet!