OPTIMAL-PSMA Trial Data Support TLX597-Tx as a Next-Generation Radioligand Therapy in Prostate Cancer
Key Takeaways
- Phase 2 OPTIMAL-PSMA dosimetry data suggest that TLX597-Tx demonstrated low salivary gland and kidney uptake in metastatic castration-resistant prostate cancer.
- This favorable profile may support dose intensification while reducing risks of xerostomia and renal toxicity.
- The findings also support further evaluation in earlier-stage metastatic hormone-sensitive prostate cancer, described in the program as androgen pathway-sensitive prostate cancer.
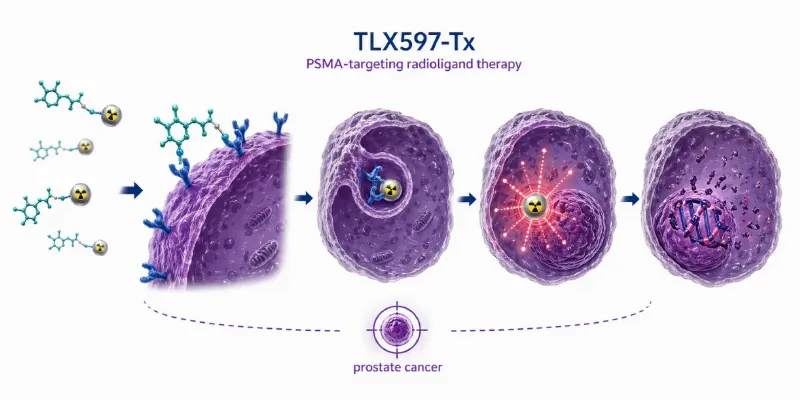
Telix Pharmaceuticals has reported initial dosimetry results from the randomized Phase 2 OPTIMAL-PSMA trial of TLX597-Tx, a next-generation PSMA-targeting radioligand therapy candidate, in men with metastatic castration-resistant prostate cancer. The data were presented at the 2026 International Prostate Cancer Symposium in Lugano, Switzerland, and suggest that TLX597-Tx may offer a wider therapeutic window by delivering high tumor radiation while limiting exposure to sensitive organs.
The findings are clinically relevant because radioligand therapy has become an important treatment approach in advanced prostate cancer, but uptake in healthy tissues, particularly the salivary glands and kidneys, can limit tolerability and complicate its use in earlier-stage metastatic disease.
Aiming to Improve Tolerability and Tumor Control
TLX597-Tx, also known as 177Lu-DOTA-HYNIC-panPSMA, is a small molecule radioligand therapy designed to target prostate-specific membrane antigen, PSMA. According to Telix, the candidate has shown low radiation uptake in healthy organs, including the salivary glands and kidneys. This profile may help reduce the risk of xerostomia, or dry mouth, and renal toxicity, two key safety concerns associated with PSMA-targeted radioligand therapy.
The company says this lower normal-organ exposure, combined with higher tumor uptake compared with existing PSMA radioligand therapies, may allow dose intensification while preserving quality of life. This is particularly important in earlier-stage metastatic prostate cancer, where patients may live longer with disease and treatment-related toxicity can have a major impact on daily function.
OPTIMAL-PSMA Tests an Intensified Dosing Strategy
OPTIMAL-PSMA is an open-label, multicenter, randomized Phase 2 investigator-initiated trial led by Professor Louise Emmett at St Vincent’s Hospital in Sydney, Australia. The study is evaluating the safety, dosimetry and efficacy of TLX597-Tx in 120 men with advanced metastatic castration-resistant prostate cancer.
Participants are randomized on a 2:1 basis to receive either an intensified dosing regimen or a standard dosing schedule. The intensified regimen delivers 8.5 GBq on day 1, day 3 and day 15, followed by dosing every 10 weeks for three additional cycles. The rationale is to increase radiation exposure to cancerous lesions at a time when they may be most vulnerable to damage, with the goal of improving response without increasing toxicity.
Professor Louise Emmett, Principal Investigator for OPTIMAL-PSMA, said, “The goal of OPTIMAL-PSMA is to identify a dose regimen for TLX597-Tx that leads to deeper and longer responses without increasing toxicity for men with metastatic prostate cancer. We look forward to starting the Phase 2 OPTIMAL-E trial soon.”
Moving Toward Earlier-Stage Metastatic Disease
Based on the dosimetry findings, Telix is planning further evaluation of TLX597-Tx in earlier-stage disease. The company is initiating OPTIMAL-E, a Phase 2 study in androgen pathway-sensitive metastatic prostate cancer, also referred to as metastatic hormone-sensitive prostate cancer.
“These encouraging dosimetry results from OPTIMAL-PSMA, combined with earlier exploratory work6, are very promising and highlight TLX597-Tx’s potential to substantially increase the tumor dose while minimizing radiation to sensitive organs. For people living with earlier-stage metastatic disease, preserving quality-of-life alongside effective cancer control is mandatory. These findings support further study in mHSPC and reinforce Telix’s strategy to develop differentiated PSMA-targeting therapies, so clinicians may be able to tailor treatment choice to the patient’s disease stage and individual condition,” said Dr. David N. Cade, Group Chief Medical Officer at Telix.
A Portfolio Approach to PSMA-Targeted Therapy
Telix is developing TLX597-Tx alongside TLX591-Tx, lutetium-177 rosopatamab tetraxetan, its lead antibody-based prostate cancer therapy candidate. TLX591-Tx is currently being studied in the Phase 3 ProstACT Global trial in metastatic castration-resistant prostate cancer.
The two programs are intended to address different clinical settings. TLX591-Tx is being developed as a radio antibody-drug conjugate in combination with standard of care for mCRPC, while TLX597-Tx is being positioned for potential use in earlier-stage metastatic hormone-sensitive prostate cancer. Both candidates remain investigational. TLX591-Tx and TLX597-Tx have not received marketing authorization in any jurisdiction.



Comments
No Comments Yet!