Phase 3 MagnetisMM-5 Trial Supports Earlier Use of Elranatamab in Relapsed Myeloma
Key Takeaways
- The Phase 3 MagnetisMM-5 trial showed that elranatamab (ELREXFIO) significantly improved progression-free survival versus daratumumab plus pomalidomide and dexamethasone in adults with relapsed or refractory multiple myeloma.
- The 497-patient study met its primary endpoint at interim analysis, with most elranatamab-treated patients remaining progression-free.
- Safety was consistent with the known profile, and no new safety signals were identified.
Pfizer has reported positive topline results from the Phase 3 MagnetisMM-5 trial, showing that elranatamab significantly improved progression-free survival in adults with relapsed or refractory multiple myeloma who had received at least one prior line of therapy.
Phase 3 Trial Meets Primary Endpoint
The randomized study compared subcutaneous elranatamab monotherapy with standard-of-care daratumumab plus pomalidomide and dexamethasone, DPd. According to Pfizer, the trial met its primary endpoint at interim analysis, demonstrating a statistically significant and clinically meaningful improvement in progression-free survival, as assessed by blinded independent central review.
The result is clinically relevant because elranatamab is already used in later-line relapsed or refractory multiple myeloma, and MagnetisMM-5 evaluates the drug in an earlier treatment setting. Pfizer said the progression-free survival benefit exceeded the pre-specified interim efficacy threshold, with most patients treated with ELREXFIO remaining progression-free at the time of analysis.
Overall survival, a key secondary endpoint, was not mature at the interim analysis. The trial is continuing to further assess survival outcomes. Pfizer said the data will be discussed with global health authorities, and detailed results will be submitted for presentation at a future medical congress.
An Earlier-Line Opportunity in Double-Class Exposed Disease
For clinicians, the MagnetisMM-5 result may be important because it addresses a double-class exposed patient population, including patients previously treated with lenalidomide and a proteasome inhibitor. This is a group in whom durable disease control can become increasingly difficult as resistance accumulates and treatment sequencing becomes more complex.
“Effective intervention earlier in the course of disease represents a critical opportunity to improve outcomes for people living with multiple myeloma,” said Jeff Legos, Chief Oncology Officer, Pfizer. “ELREXFIO has already helped address a significant unmet need in heavily pre‑treated patients, delivering deep, durable, and clinically meaningful responses. The MagnetisMM-5 results reinforce our confidence in ELREXFIO’s potential to benefit patients earlier in their treatment journey and support our comprehensive strategy to evaluate ELREXFIO both as monotherapy and as part of combination approaches across multiple lines of therapy.”
MagnetisMM-5 Trial Design and Dosing Strategy
MagnetisMM-5 is an open-label, multicenter, randomized Phase 3 study that enrolled 497 patients across 26 countries. Eligible patients had relapsed or refractory multiple myeloma and had received at least one previous line of therapy, including lenalidomide and a proteasome inhibitor. Patients assigned to elranatamab received two step-up priming doses followed by a weekly 76 mg subcutaneous injection. Treatment frequency could be reduced to every two weeks after 24 weeks in patients who achieved a partial response for more than two months, and then to every four weeks for all patients after 48 weeks on therapy.
Safety Profile Remains Consistent
The safety and tolerability profile of elranatamab in MagnetisMM-5 was consistent with the known profile of the drug, and no new safety signals were identified. This is particularly relevant for bispecific antibodies, where careful monitoring for immune-mediated toxicities, infections, and treatment burden remains an important part of clinical use.
How Elranatamab Redirects T Cells Against Myeloma
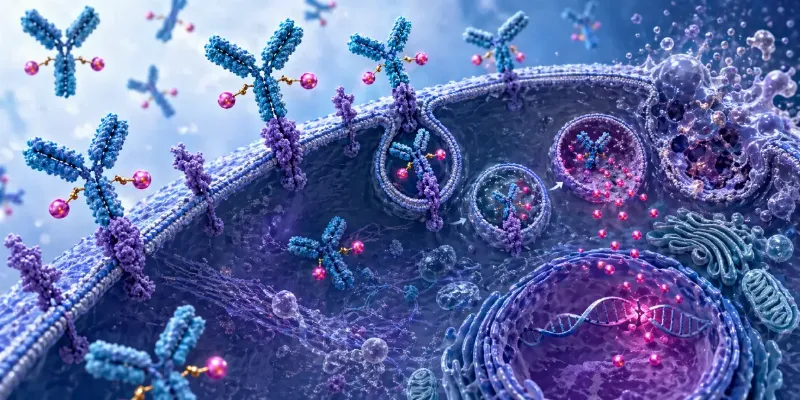
ELREXFIO is a BCMA-CD3 bispecific antibody designed to bind B-cell maturation antigen on myeloma cells and CD3 on T cells, thereby redirecting T-cell activity against malignant plasma cells. The agent is administered subcutaneously and is being evaluated across multiple lines of therapy, both as monotherapy and in combination regimens, as part of Pfizer’s MagnetisMM clinical development program.
Elranatamab (ELREXFIO) is approved in more than 35 countries. In the United States, it received accelerated approval for adults with relapsed or refractory multiple myeloma who have received at least four prior lines of therapy, including a proteasome inhibitor, an immunomodulatory agent, and an anti-CD38 monoclonal antibody. In the European Union, it has conditional marketing authorization for adults with relapsed or refractory multiple myeloma who have received at least three prior therapies and whose disease progressed on the last therapy.
MagnetisMM-5 is part of a broader development strategy in double-class exposed multiple myeloma. Pfizer is also conducting MagnetisMM-32, a fully recruited Phase 3 trial evaluating elranatamab in patients previously treated with daratumumab as part of standard front-line therapy.



Comments
No Comments Yet!